| J Mult Scler Neuroimmunol > Volume 14(1); 2023 > Article |
|
ABSTRACT
Immune-related neuromuscular complications due to immune checkpoint inhibitors (ICIs) are widely reported. However, cases involving vasculitic neuropathy (VN) are rare. We report a case of pembrolizumab-associated VN, confirmed by nerve biopsy, presenting as mononeuritis multiplex with erythematous skin lesions. Based on negative serological tests for other causes of vasculitis and temporal relationship with ICI treatment, vasculitis was suspected to be associated with pembrolizumab. VN should be considered in patients with progressive neurological symptoms and skin lesions after receiving ICI therapy.
Pembrolizumab is an immune checkpoint inhibitor (ICI) that selectively blocks programmed death receptor 1 (PD-1) and enhances T-cell activation and anti-tumor responses.1 Although pembrolizumab is widely used in advanced stage cancers, various neuromuscular immune-related adverse events (irAEs), including myositis, myasthenia gravis, and Guillain-Barré syndrome, are known to occur.2,3 Vasculitis is also a well-known irAE of ICI treatment.4,5 However, vasculitic neuropathy (VN) induced by ICI therapy is rare.3,6,7 Herein, we present a case of pembrolizumab-associated VN.
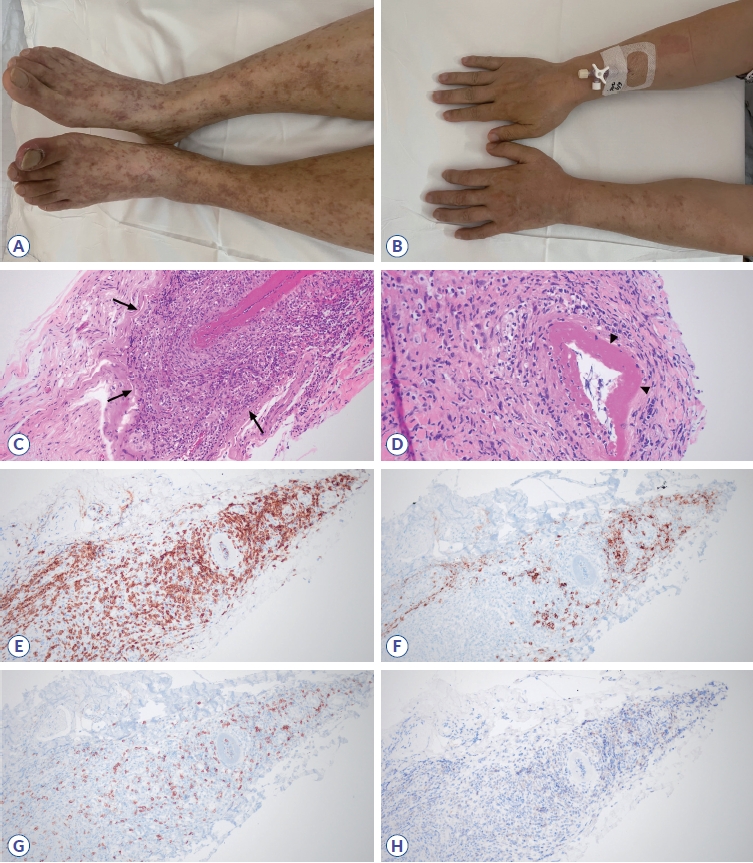
A 57-year-old male presented with erythematous skin lesions on upper and lower extremities that advanced to painful tingling sensation, numbness, and weakness in the fourth and fifth fingers of his left hand after his ninth cycle of pembrolizumab therapy. He had a history of hypertension and was receiving pembrolizumab for stage IV non-small cell lung cancer. The numbness started in both feet after the sixth cycle of pembrolizumab therapy, simultaneous with the onset of skin lesions. Reticulated dark erythematous patches were present on both arms and legs (Fig. 1A, B). Neurological examination showed a claw hand feature with Medical Research Council (MRC) grade 4 weakness in left fourth and fifth finger flexion and abduction and hypoesthesia in the medial aspect of the left hand.
Three weeks after the visit, the patient additionally complained of pain and weakness in the first and second fingers on his left hand and of a painful tingling sensation and weakness in his left foot. Neurological examination revealed weakness in abduction of the left thumb (MRC grade 2), abduction of the second and fifth fingers (MRC grade 2), dorsiflexion and plantarflexion of the left ankle (MRC grade 4), and hypoesthesia in his left fingers and palm and in both the plantar side and dorsum of the left foot.
Nerve conduction study showed conduction block in the left median and left peroneal nerves and reduced amplitude of compound muscle action potentials in the left ulnar and right tibial nerves. Amplitude of sensory nerve action potentials was reduced in the left median, ulnar, superficial peroneal, and sural nerves.
Serological tests for systemic vasculitis, including tests for antinuclear antibodies (Abs), antineutrophil cytoplasmic Abs, anti-glomerular basement membrane Abs, and anti-C1q Abs, were all negative. Tests for paraneoplastic Abs including anti-Hu, Yo, Ri, CV2, Ma2/Ta, amphiphysin, recoverin, and SOX1 were negative. Tests for Abs against human immunodeficiency virus, hepatitis B surface antigen, and hepatitis C virus were also negative. Biopsy of the skin lesions showed lymphocytic vasculitis. Biopsy of the left sural nerve showed prominent leukocytoclastic vasculitis, and immunohistochemical staining revealed inflammatory cell infiltrations, especially CD8+ T-cells (Fig. 1C-H). Based on the diagnosis of pembrolizumab-associated VN, pembrolizumab was discontinued and intravenous methylprednisolone therapy (1 g for 5 days) followed by 1 mg/kg of oral prednisolone was administered. After 5 weeks of steroid treatment, skin lesions improved, and no further progression of peripheral neuropathy was reported. However, significant improvement in weakness was not observed, and the patient was lost to follow-up after this visit.
ICI-associated vasculitis occurs approximately 3 months (range, 1.2-6 months) after onset of ICI therapy.4 Although vessels of all sizes are involved, frequently reported types of ICI-associated vasculitis include giant cell arteritis (GCA) and primary angiitis of the central nervous system, accounting for 15% and 20% of cases, respectively.4 GCA is thought to be induced by infiltration of CD4+ T-cells and macrophages into vessel walls. Cases of VN associated with peripheral neuropathy have rarely been reported. Kao et al.3 previously reported a case of asymmetric VN induced by nivolumab, a PD-1 inhibitor. Nivolumab was used to treat stage IV peritoneal mesothelioma, and vasculitis occurred after the fifth cycle.1 In two additional cases, pembrolizumab-induced VN reportedly developed 2 weeks after the first infusion and after 10 cycles of pembrolizumab treatment.6,7 Nerve biopsy in both previous and present cases of pembrolizumab-induced VN showed marked CD8+ T-cell infiltration in contrast to a previously reported case of GCA that mainly showed CD4+ T-cell infiltration.7 Although mechanisms of ICI-related VN are poorly understood, we hypothesize that CD8+ T-cells play a major role in vasculitis affecting small vessels.
In the present case, the underlying cause of progressive weakness, sensory loss, and erythematous skin lesions was pathologically confirmed as vasculitis. Initial symptoms occurred approximately 4 months after ICI therapy in the present case, which is in the range of the previously reported period. Based on the temporal relationship between symptoms and ICI treatment as well as lack of evidence of other conditions such as systemic vasculitis or paraneoplastic syndrome, vasculitis was suspected to be associated with pembrolizumab therapy. Although extremely rare, VN should be considered in patients presenting with progressive weakness, sensory changes, and skin lesions. Early diagnosis and appropriate treatment can reduce neurological sequelae.
Figure 1.
Skin lesions and histopathological results of the left sural nerve biopsy. Reticulated erythematous skin lesions on both legs (A) and arms (B). Hematoxylin and Eosin staining shows prominently inflamed blood vessels (C, arrows, ×200) and marked fibrin deposition in the vessel wall (D, arrowheads, ×400), representing leukocytoclastic vasculitis. Immunohistochemical (IHC) staining demonstrates prominent T-cell infiltration (E, red, ×200) compared with B-cells (F, ×200). Infiltration of CD8+ T-cells (G, ×200) were prominent to CD4+ T-cells (H, ×200).
REFERENCES
1. Khoja L, Butler MO, Kang SP, Ebbinghaus S, Joshua AM. Pembrolizumab. J Immunother Cancer 2015;3:36.



2. Möhn N, Beutel G, Gutzmer R, Ivanyi P, Satzger I, Skripuletz T. Neurological immune related adverse events associated with nivolumab, ipilimumab, and pembrolizumab therapy-review of the literature and future outlook. J Clin Med 2019;8:1777.



3. Kao JC, Liao B, Markovic SN, Klein CJ, Naddaf E, Staff NP, et al. Neurological complications associated with anti-programmed death 1 (PD-1) antibodies. JAMA Neurol 2017;74:1216-1222.



4. Daxini A, Cronin K, Sreih AG. Vasculitis associated with immune checkpoint inhibitors-a systematic review. Clin Rheumatol 2018;37:2579-2584.


5. Boland P, Heath J, Sandigursky S. Immune checkpoint inhibitors and vasculitis. Curr Opin Rheumatol 2020;32:53-56.


-
METRICS

-
- 0 Crossref
- 0 Scopus
- 1,564 View
- 30 Download
- Related articles



 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print